Basic Information
| Drug ID | DDPD00104 |

|
| Drug Name | Octreotide | |
| Molecular Weight | 1019.25 | |
| Molecular Formula | C49H66N10O10S2 | |
| CAS Number | 83150-76-9 | |
| SMILES | [H][C@]1(NC(=O)[C@H](CCCCN)NC(=O)[C@@H](CC2=CNC3=C2C=CC=C3)NC(=O)[C@H](CC2=CC=CC=C2)NC(=O)[C@H](CSSC[C@H](NC1=O)C(=O)N[C@H](CO)[C@@H](C)O)NC(=O)[C@H](N)CC1=CC=CC=C1)[C@@H](C)O | |
| External Links | ||
| DRUGBANK | DB00104 | |
| PubChem Compound | 448601 | |
| PDR | 438 | |
| Drugs.com | Drugs.com Drug Page | |
Experimental Physicochemical Property
| Property Name | Property Value | Unit | Raw Value | Raw Unit | Reference |
|---|---|---|---|---|---|
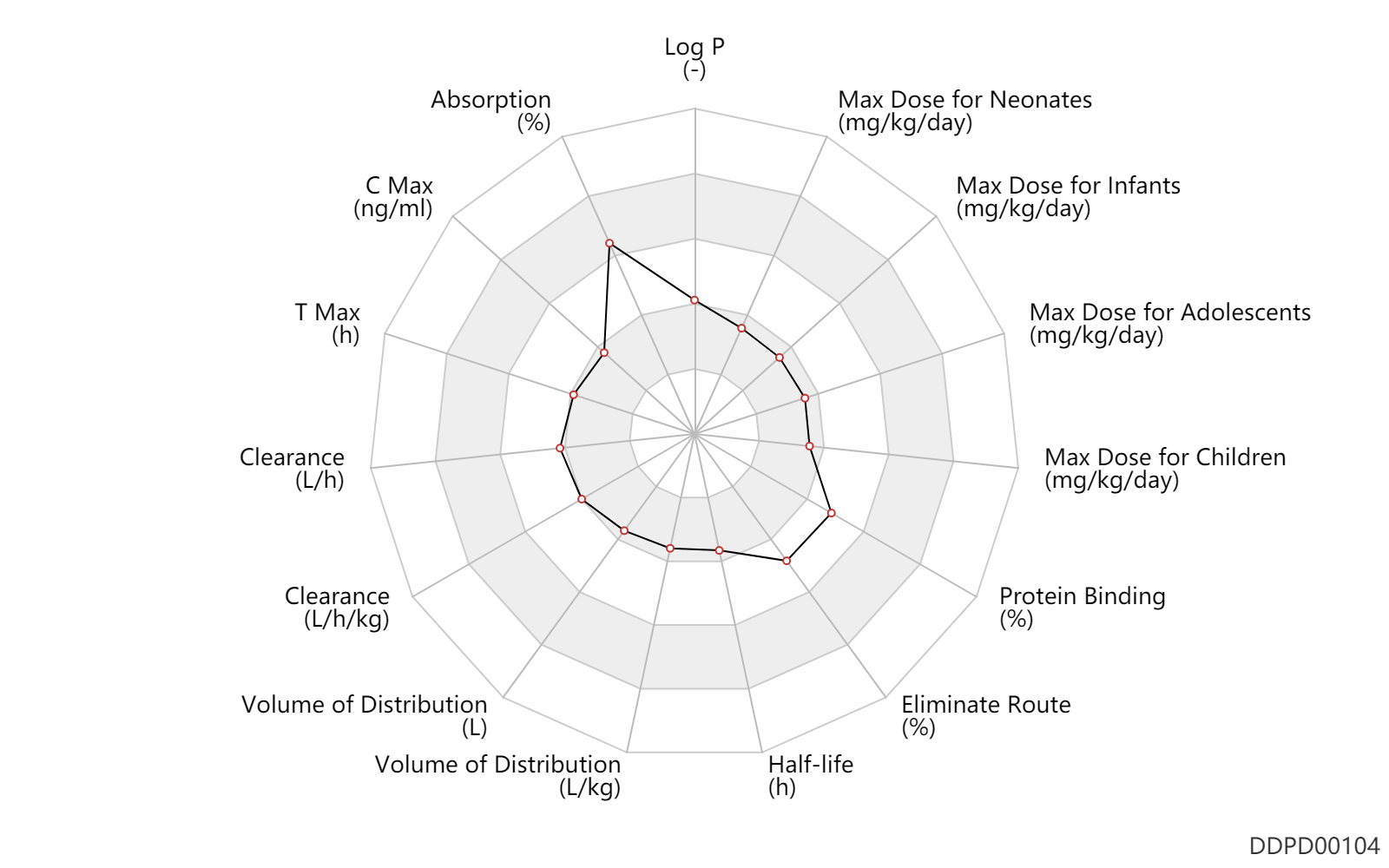
| Log P | 1.0 | - | 1.0 | - | DRUGBANK |
Pharmacokinetic/ Toxicokinetic Property
| Property Name | Property Value | Unit | Raw Value | Raw Unit | Annotation | Reference |
|---|---|---|---|---|---|---|
| Absorption | 100.0 | % | ~100 | % | subcutaneous injection, SC; | DRUGBANK |
| C Max | 2.5 | ng/ml | 2.5 | ng/ml | Capsule, PO, Oral; acromegaly; | DRUGBANK | C Max | 5.3 | ng/ml | 5.3 | ng/ml | Capsule, PO, Oral; acromegaly; | DRUGBANK |
| T Max | 0.50 | h | 0.5 | h | subcutaneous injection, SC; | DRUGBANK | T Max | 2.1 | h | 1.67-2.5 | h | Capsule, PO, Oral; | DRUGBANK |
| Clearance | 8.5 | L/h | 7.0-10 | L/h | DRUGBANK | Clearance | 0.14 | L/h/kg | 2.38 | ml/min/kg | intravenous injection, IV; human, homo sapiens; | Human Intravenous Pharmacokinetic Dataset |
| Volume of Distribution | 13.6 | L | 13.6 | L | normal,healthy; | DRUGBANK | Volume of Distribution | 24.3 | L | 18.1-30.4 | L | intravenous injection, IV; | DRUGBANK | Volume of Distribution | 0.19 | L/kg | 0.19 | L/kg | intravenous injection, IV; human, homo sapiens; | Human Intravenous Pharmacokinetic Dataset |
| Half-life | 0.20 | h | 0.2 | h | elimination half-life; subcutaneous injection, SC; | DRUGBANK | Half-life | 2.5 | h | 2.3-2.7 | h | elimination half-life; PO, oral; subcutaneous injection, SC; | DRUGBANK | Half-life | 1.5 | h | 72-113 | min | elimination half-life; PO, oral; | DRUGBANK | Half-life | 1.7 | h | 1.7 | h | intravenous injection, IV; human, homo sapiens; | Human Intravenous Pharmacokinetic Dataset |
| Eliminate Route | 32.0 | % | 32 | % | Urinary excretion; | DRUGBANK | Eliminate Route | 32.0 | % | ~32 | % | Urinary excretion; Unchanged drug; | DRUGBANK |
| Protein Binding | 65.0 | % | ~65 | % | DRUGBANK |
Maximum Dosage
| Property Name | Property Value | Unit | Raw Value | Raw Unit | Annotation | Brand Name | Component | Reference |
|---|---|---|---|---|---|---|---|---|
| Max dose for neonates | 0.24 | mg/kg/day | 10 | mcg/kg/hour | intravenous infusion, iv in drop | Sandostatin | octreotide acetate | PDR |
| Max dose for neonates | 0.04 | mg/kg/day | 40 | mcg/kg/day | subcutaneous injection, SC;intravenous injection, IV; | Sandostatin | octreotide acetate | PDR |
| Max dose for infants | 0.24 | mg/kg/day | 10 | mcg/kg/h | intravenous infusion, iv in drop | Sandostatin | octreotide acetate | PDR |
| Max dose for infants | 0.04 | mg/kg/day | 40 | mcg/kg/day | subcutaneous injection, SC;intravenous injection, IV; | Sandostatin | octreotide acetate | PDR |
| Max dose for children | 0.24 | mg/kg/day | 10 | mcg/kg/hour | intravenous infusion, iv in drop | Sandostatin | octreotide acetate | PDR |
| Max dose for children | 0.04 | mg/kg/day | 40 | mcg/kg/day | subcutaneous injection, SC;intravenous injection, IV; | Sandostatin | octreotide acetate | PDR |
| Max dose for children | 0.015 | mg/kg/day | 15 | mcg/kg/day | subcutaneous injection, SC | Sandostatin | octreotide acetate | PDR |
| Max dose for adolescents | 0.015 | mg/kg/day | 15 | mcg/kg/day | subcutaneous injection, SC | Sandostatin | octreotide acetate | PDR |